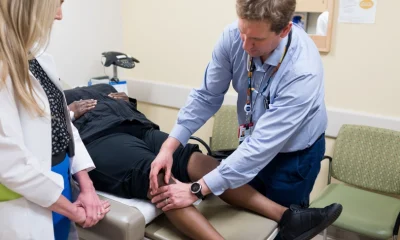
Health
Applications open for health care careers scheme for displaced people in Wales

Programme offers pathway into NHS roles
APPLICATIONS have opened for the Royal College of Nursing’s health care careers programme, aimed at supporting refugees and asylum seekers living in Wales to gain practical experience and insight into NHS roles.
Following the success of the first cohort earlier this year, the RCN will run a second programme between February and June 2026. It is funded by Health Education and Improvement Wales and is open to anyone living in Wales who has been displaced at any point, including refugees, asylum seekers and those with other protection backgrounds.
Practical NHS experience
The scheme is designed to help participants build confidence, resilience and communication skills while introducing them to the wide range of careers available across the Welsh health sector.
It includes practical sessions such as basic life support, manual handling, infection control and observational work experience. Participants will also receive coaching for a simulated NHS interview, along with help drafting personal statements and CVs.
Options are available to study either online or in person.
‘A workforce that reflects the population it serves’
Lorna Mayles, Head of Students and RCN King’s Nursing Cadets, said: “It’s important that the workforce of our NHS reflects the population it serves, and this project aims to introduce displaced people in Wales to the diverse roles across the health care landscape to encourage them to consider a career in health care.
“The programme is an opportunity to gain an insight into the NHS, learn new skills and get support to apply for a role in the NHS.”
How to apply
Applications are open until 6 January 2026.
For more information visit the RCN’s “Health Care Careers for Displaced People” webpage.
To apply, email [email protected]
.
Charity
Charity campaign showcases powerful bereavement support for local families

HYWEL DDA Health Charities, the official charity of Hywel Dda University Health Board, is calling on local communities to help them provide comfort and support to families who have lost a child.
The charity is running a campaign this spring to highlight its Wish Fund which provides support to families during times of bereavement.
The Wish Fund supports Hywel Dda’s Paediatric Palliative Care team to provide personalised support to families of children and young people receiving palliative care, and helps them create treasured memories when their child sadly passes away. From hand moulds and fingerprint jewellery to memory boxes and specialist sibling support, the fund ensures bereaved families are not alone during the most devastating moments of their lives.
Frankie’s parents Phil and Veronica shared how the Wish Fund helped them during their darkest days:
“We had the absolute pleasure of looking after the most beautiful, awesome, wonderful, loving and brave little lad who was very poorly in the last years of his life.

“We met his Paediatric Palliative Care team two years before his passing and boy what an awesome team they are.
“We will never be able to thank the Wish Fund enough. Thanks to the Wish Fund, we had the most amazing mementos, a Christmas bauble, a necklace with Frankie’s fingerprint on it and a mould of his hand. Those simple objects are our most treasured possessions along with a lock of his beautiful hair.”
The Wish Fund relies on charitable donations, and every contribution can have a lasting impact:
- £15 provides a teddy for a bereaved sibling
- £30 funds a sibling support session
- £50 creates a hand mould keepsake
- £70 provides a Memory Box
- £85 funds bespoke hand‑printed jewellery
These meaningful items offer comfort to parents, carers and siblings who are navigating unimaginable loss.
Rebecca McDonald, Clinical Nurse Specialist – Paediatric Palliative Care, said: “I want to extend my heartfelt thanks to everyone who has donated and continues to donate to the Wish Fund.
“Thanks to your kindness and generosity, we have been able to provide meaningful memory-making items and bereavement materials. These resources make an immeasurable difference in helping us support our families through the most difficult time.
“Your compassion and support truly help us bring comfort, dignity, and lasting memories to the families we care for. Thank you for your support in helping us make a big difference to families we support across our health board.”
To find out more or to donate to the Wish Fund, please visit: https://hywelddahealthcharities.nhs.wales/campaigns/the-wish-fund/
Business
Narberth Kadinsky gallery to dental surgery refused

PLANS to convert a former art gallery to a dental surgery on the edge of a Pembrokeshire town have been refused.
In an application to Pembrokeshire County Council, Ahmed Abouserwel, through agent A.D Architectural Design Consultants LTD, sought permission for a change of use of the former Kadinsky gallery, Redstone Road, Narberth, to a dental surgery, along with associated works.
A supporting statement said: “The existing open plan gallery space will be transformed into the main dentist area, with a glazed internal lobby, leading directly into the open reception / waiting area. There will be five treatment rooms accessed directly off the reception, with a private archive room behind the reception desk.
“The rear lean-to projection will be extended to the north to accommodate a proposed decontamination room and to re-model the Staff area and W.C provision (number to remain as existing).”
It said the proposal would create 10 full and three part-time jobs.
An officer report recommending refusal said concerns were raised by the county Highways authority, who having assessed the application on safety, capacity and policy considerations, recommended the application be REFUSED on the grounds of insufficient evidence provided.
“The submitted design and access statement and block plan indicate on-site parking provision for 16 vehicles, located to the north and west of the building. The application form states that the site will employ 10 full-time staff and three part-time staff. However, the submission does not differentiate between practitioners and ancillary/support staff.”
It said, on planning guidance, health centres require three spaces per practitioner; and one space per three ancillary staff, adding: “As the applicant has not provided a breakdown of staff roles, the Highway Authority is unable to assess whether the proposed parking provision is adequate.”
It stressed: “Whist there is no in-principle objection to the redevelopment of this established site for a dental surgery, insufficient information has been provided to fully assess the proposal.”
It was refused on the grounds including it would lead “to the unjustified loss of an employment premises in a location which contributes to the local supply of employment land and buildings,” adding: “Insufficient evidence has been submitted to demonstrate that the building is no longer suitable or viable for continued employment use, nor that there is overriding community need to justify its loss.”
It was also refused on the grounds that “Insufficient information has been submitted to demonstrate that the development would operate without giving rise to unacceptable highway safety impacts or on street parking pressure”.
Health
Patients in Wales waiting years for autoimmune diagnosis
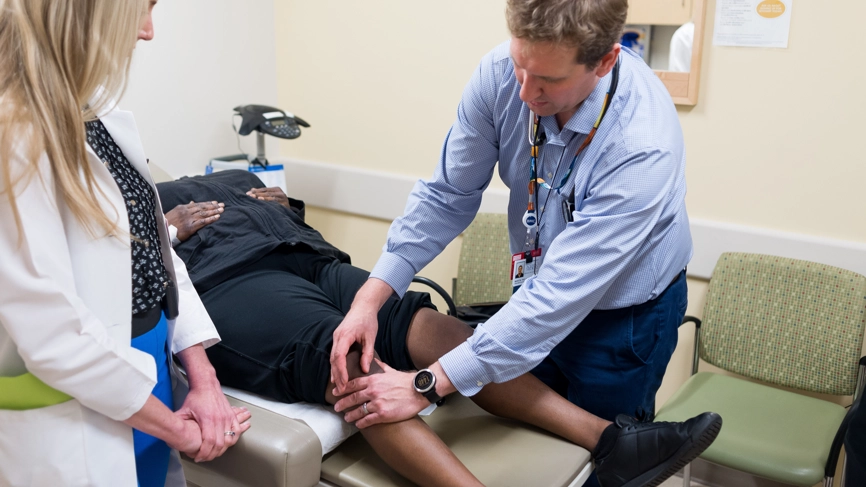
Study led by Swansea University says long delays, limited specialist access and referral barriers are leaving some patients worse off than those elsewhere in the UK
PEOPLE in Wales with autoimmune and rheumatological conditions are facing long waits for diagnosis, poor access to specialist care and repeated obstacles when trying to secure treatment outside their local area, according to new research.
The study, led by Swansea University, examined the experiences of patients with illnesses including lupus and other autoimmune disorders. It drew on interviews, survey findings and Freedom of Information responses from health boards across Wales.
Researchers found that specialist provision remains limited in some parts of the country. Some patients reported having no access to a lupus specialist in their area, while FOI responses indicated there is only one NHS neuro-ophthalmologist in Wales for patients with neurological or visual problems linked to autoimmune disease.
The report also highlighted a lack of diagnostic services. None of the Welsh health boards that responded said they currently provide nailfold capillaroscopy, a test used to help identify conditions such as scleroderma.
Diagnosis in rheumatology can also be difficult because many of the conditions involved are complex and may develop gradually over time. Illnesses such as ankylosing spondylitis can be mistaken in the early stages for more common causes of pain, while autoimmune disorders often present with symptoms that overlap with other conditions.
Patients may require a combination of investigations before a diagnosis can be confirmed, including HLA-B27 testing, routine blood work, MRI scans, X-rays and detailed clinical assessment. Doctors also rely heavily on patient interviews to build up a picture of pain, stiffness, mobility and the wider pattern of symptoms.
There have also been local pressures on services in west Wales. At Withybush Hospital, delays were caused after the departure of a specialist consultant, and it took some time for that post to be filled. That consultant has now been replaced. However, it is understood the workload remained heavy, with the specialist working through a backlog of cases.
In some cases, patients may also feel rheumatology is treated as a lower-profile service. At Withybush Hospital, for example, the department has been based in a portacabin in the hospital car park, which it shares with the lymphoedema service — a setting that can give the impression that two important but often overlooked specialties are being accommodated on the margins of the wider hospital system.
Rheumatology also places a significant burden on the NHS because of the cost of long-term treatment and follow-up care, although that is not the main focus of the study. Biological drugs used to treat conditions such as rheumatoid arthritis and ankylosing spondylitis can cost around £500 for a pack of four injections, according to local hospital sources. Patients on those medications often also require regular blood monitoring, consultant reviews, specialist nurse support and physiotherapy. The Herald understands that more than 1,000 patients in Pembrokeshire are currently being managed through this treatment pathway.
Researchers said patients were also being held back by the referral system. Under current NHS Wales rules, doctors must apply to their local health board for permission to send patients for treatment outside the area or across the border into England. The study said decision-making around those requests was often inconsistent and unfair.
One patient with systemic lupus erythematosus told researchers he waited two years for approval to be referred to England, despite believing the process should only have taken weeks.
The study found that patients in Wales often face longer journeys to diagnosis than elsewhere in the UK. In the sample reviewed by researchers, the average time from first symptoms to diagnosis was around 11 years, compared with around seven years across the UK.
Patients described the effect those delays had on their health. One woman in her 30s said requests to see a specialist in a neighbouring health board had been turned down, and that her lupus had worsened as a result. Another patient said access to care became more difficult after moving from England to Wales.
The research was led by Rupert Harwood of Swansea University Medical School, who himself lives with several autoimmune conditions.
His own experience reflects the wider concerns raised in the report. After developing visual symptoms in 2016, he was advised to see a neuro-ophthalmologist, but no such service was then available in Wales. A referral to England was not successfully made until 2025, and he is still waiting for an appointment.
Mr Harwood said the present system can leave patients at a disadvantage if they cannot afford to seek private treatment outside Wales.
The study concludes that while NHS Wales rheumatology teams provide high-quality care, patients in Wales face additional barriers compared with those in England.
Researchers are calling for a review of the out-of-area referral system, better access to specialist services and greater investment in diagnostic testing to reduce delays and improve outcomes.
-

 Business7 days ago
Business7 days agoA proper Pembroke local: Inside the warm welcome of The Old Cross Saws Inn
-

 Business5 days ago
Business5 days agoMilford Haven dry dock pitched as home for giant £480m superyacht yard
-

 Community7 days ago
Community7 days agoDoorstep warning issued after reports of suspicious callers in Pembrokeshire
-

 Crime7 days ago
Crime7 days agoRomino’s licence revoked after illegal workers found and fire risks exposed
-

 Politics6 days ago
Politics6 days agoWelsh Lib Dems promise childcare boost, social care reform in manifesto launch
-

 News6 days ago
News6 days agoHaverfordwest County AFC escapes winding-up order but ordered to pay costs
-

 News6 days ago
News6 days agoLabour split over Brawdy defence project as Eluned Morgan calls for project halt
-

 Community6 days ago
Community6 days agoFamily safe after major house fire in Letterston, community told